
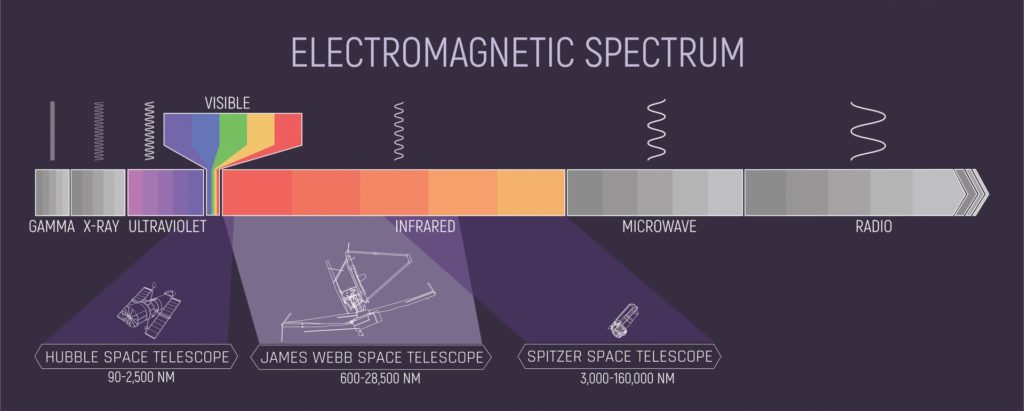
Finding an earthlike planet or one that could be inhabited is possible. The James Webb Space Telescope is equipped with near-infrared (NIR) cameras and spectrometers that are already helping astronomers peer into the chemical composition of distant planets. In the near-infrared region of the electromagnetic spectrum, many molecular signatures are detected. Spectroscopic techniques can identify the atmospheric composition of planets as they move around their star. Their sun will act as a light source that will shine through the planet’s atmosphere. The resulting light is then compared to the energy before and after the planet moves through the light shining toward us.
The exoplanet’s atmospheric molecules leave their molecular signature in the near-infrared regions of the electromagnetic spectrum where the telescope spectrometers are tuned. NASA has a great explanation of this process on its website.1 The molecular signatures are extremely weak, but many molecules are discernible with repeated measurements, and all of this data should allow scientists to determine which molecules are present and the relative amounts of each. Reports are already being published. For example, carbon dioxide was identified in the atmosphere of the exoplanet WASP-39b using the telescope.2

Which molecules can the telescope detect? The list includes such molecules as water, carbon dioxide, carbon monoxide, methanol, ammonia, and methane. This is a very interesting list of atmospheric molecules that seemingly give a sense of a planet’s habitability. What might be overlooked is that these molecules are all infrared-active, which makes these good greenhouse gases. This is not a bad thing, but the spectrometers on the telescope cannot pick up other important life-permitting molecules such as dioxygen and dinitrogen. Detecting water in the atmosphere of distant planets is right in the wheelhouse of this new telescope, and many scientists are trying to book time on the telescope to assist in this detection process.3 The information relative to the water content of alien atmospheres will be extremely helpful in understanding the prevalence of water throughout the cosmos. A recent study of the atmosphere of exoplanet VHS 1256-1257 b suggests water is present along with silicates, methane, carbon monoxide, and carbon dioxide.4 Could this be a habitable place for life?
When it comes to determining if these planets are habitable, a whole host of chemical factors need to be considered, and the telescope will provide some of these details. Unfortunately, it will not provide a complete picture. From most reports, the general sense of a planet’s habitability is that it resides in the habitable zone, which allows liquid water to exist. However, a planet needs more than just liquid water. I would suggest that the presence of dioxygen and dinitrogen are just as important. These gases can support complex life along with a small, variable amount of greenhouse gases to heat the planet.
The planet cannot have too many greenhouse gases, since many of them are toxic or too heavy, and they could displace oxygen and suffocate any inhabitants. A habitable planet needs oxygen and nitrogen in nearly the same ratios found on earth. Oxygen is critical for complex and larger life forms to exist.5 It is possible that some microbes could exist on planets without oxygen, but creatures bigger than unicellular anaerobes require dioxygen to power their metabolism.
Why is dinitrogen so critical? In my study I have identified at least 10 parameters that make dinitrogen the ideal partner for dioxygen. Observing dinitrogen along with dioxygen in the atmospheres of potentially habitable planets will provide a more definite picture. Unfortunately, these diatomic molecules have their spectral signatures in the X-ray and UV region of the electromagnetic spectrum, which is not part of the telescope’s instrumentation. The planet’s atmosphere is responsible for maintaining liquid water with about 15 psi (pounds per square inch) of pressure. Dioxygen makes up about 3 psi, but the remaining 12 psi needs to come from a diatomic molecule that is similar to oxygen’s density and must have the following attributes:
» unreactive to oxygen
» nonflammable
» transparent in the visible/infrared
» nongreenhouse-forming
» some ability to absorb harmful short wavelength radiation (UV, X-ray, and gamma ray)
» nonreactive to life (inert)
» minimally soluble in water
» nonacidifying in water
» useful for life
No other molecule matches these criteria as precisely as dinitrogen. The total air pressure is critical, as that sets the proper pressure to maintain water as a liquid. Too little pressure, and water will evaporate into the air, causing an increase in humidity, global warming, and removal of water from lakes, rivers, and organisms. Too much air pressure will effectively shut down the water cycle that cleanses and desalinates water for reuse. To find a habitable planet for humans, earth’s atmosphere sets the standard for many chemical reasons.
The local star near an exoplanet also needs to be a good match that resembles our own sun in terms of its spectral output. If the star produces too much energy in the infrared or near-infrared range, this light does not have sufficient energy to drive photosynthesis and other life-permitting chemical reactions. If the star produces too much energy in the UV range (or X-ray and gamma ray), this will destroy any carbon-based molecules, which is the only type of atom that meets the fitness requirements for life.6
The number of parameters is quite long, making earth a rare exception in the universe, according to some scientists.7 The telescope expands our view and understanding of the chemical composition of exoplanet atmospheres. All of this new information will help us understand how common certain atmospheric molecules are in the universe. The scientific community will get a better understanding of the probability of finding an earthlike planet. But will we find earth’s equal? The telescope is a nice step forward in this search, but the probability seems quite low, given all of the requirements. Until we can get a complete chemical profile of an exoplanet’s atmosphere, looking for water and the other infrared-active gases will be a nice step forward, but it will fall far short.
What does Scripture say about other habitable planets? Is there a chance that some worlds could support life? Some have suggested that the “sons of God,” mentioned in Job 1:6, 7, who visit in heaven could indicate other planets with human-like inhabitants. From Ellen White’s writings, we know that other inhabited worlds do exist but are beyond our reach. In Early Writings White writes, “Then the angel said, ‘You must go back, and if you are faithful, you, with the 144,000, shall have the privilege of visiting all the worlds and viewing the handiwork of God.’ ”8 I firmly believe there are other habitable planets, but they exist by God’s design and only by His handiwork. The telescope will help determine the possibility of a habitable planet, and this new information will help the world see the might and power of our Creator’s hand.
1 C. Pulliam, “NASA’s Webb Space Telescope to Inspect Atmospheres of Gas Giant Exoplanets,” NASA.gov, July 11, 2018, https://www.nasa. gov/feature/goddard/2018/nasa-s-webb-space-telescope-to-inspect-atmospheres-of-gas-giant-exoplanets.
2 T. Pultarova, “James Webb Space Telescope Sniffs Out Carbon Dioxide Around an Alien World,” Space.com, 2022, https://www.space.com/ james-webb-space-telescope-exoplanet-carbon-dioxide.
3 K. Cooper, “Possible Water World Spotted Orbiting an Alien Star,” Space.com, 2022, https://www.space.com/ ocean-world-habitable-zone-potential-detection.
4 B. E. Miles et al., “The TELESCOPE: Early Release Science Program for Direct Observations of Exoplanetary Systems II: A 1 to 20 Micron Spectrum of the Planetary-Mass Companion VHS 1256-1257 b,” 2022, arXiv:2209.00620v1.
5 N. Lane, Oxygen: The Molecule That Made the World (Oxford: Oxford University Press, 2002).
6 M. Denton, Nature’s Destiny: How the Laws of Biology Reveal Purpose in the Universe (New York: Free Press, 2002).
7 See, for example, G. Gonzalez and J. W. Richards, The Privileged Planet: How Our Place in the Cosmos Is Designed for Discovery (Washington, D.C.: Gateway Editions, 2020) and P. D. Ward and D. Brownlee, Rare Earth: Why Complex Life is Uncommon in the Universe (New York: Springer, 2000).
8 Ellen G. White, Early Writings (Washington, D.C.: Review and Herald Pub. Assn., 1882, 1945), p. 40.